Key mechanism in essential enzyme uncovered
Scientists know that when certain steroids bind to the essential enzyme known as the sodium pump, it can both kill and cure. Now they have also solved the mystery of how this molecular binding occurs.
The heart is pumping. You’re alive. But if you eat certain plants such as the foxglove (Digitalis purpurea), your heart may well stop beating.
The plant contains cardiotonic steroids that are so toxic that they can kill you, but they can also save your life. If the steroids are dosed correctly, they have a healing effect on diseases such as cardiovascular disease, hypertension and certain types of cancer. This is why it has been used as heart medication for 200 years.
Arrows with toxic steroids
Now, for the first time, scientists have described in atomic detail how the cardiotonic steroids work by binding to an essential enzyme in the body. This is the first and crucial step towards understanding how the steroids can not only kill, but can also get a sick and weak heart to start beating rhythmically again.
“These steroids are very dangerous substances. They are used as defensive weapons by animals and plants, and in Africa they have been used as arrow poison. We know that the steroids are found in plants, reptiles and that even the human body produces them,” says Associate Professor Natalya Fedosova of the Department of Biomedicine and the PUMPkin centre of excellence at Aarhus University, Denmark.
“However, when dosed correctly the steroids have a healing effect on cardiovascular disease, hypertension, some types of cancer and a string of other diseases. That’s why they’re used as heart medication, and tests are currently underway to develop cancer medication.”
Steroids bind to sodium pumps
It has long been known that cardiotonic steroids bind to an essential enzyme, which was originally discovered by the Danish professor Jens Christian Skou. In 1997 he received the Nobel Prize in Chemistry for discovering the enzyme, which is found in all cells in our body.
This enzyme is known as the Na+/K+-ATPase (known in daily speech as the sodium pump), because it ensures that sodium ions (Na+) are constantly pumped out of the body’s cells and that potassium ions (K+) are pumped in.
Watch an explanation of how the sodium pump works in this Youtube Video.
Important description of molecular binding
Scientists know about the function and structure of the sodium pump, and numerous scientific articles have been published about the cardiotonic steroids, which have been used as medication for centuries.
Exactly how the steroids bind to and affect the sodium pump has, however, so far remained a mystery. But now the research team from Aarhus University has managed to get a clear picture of it.
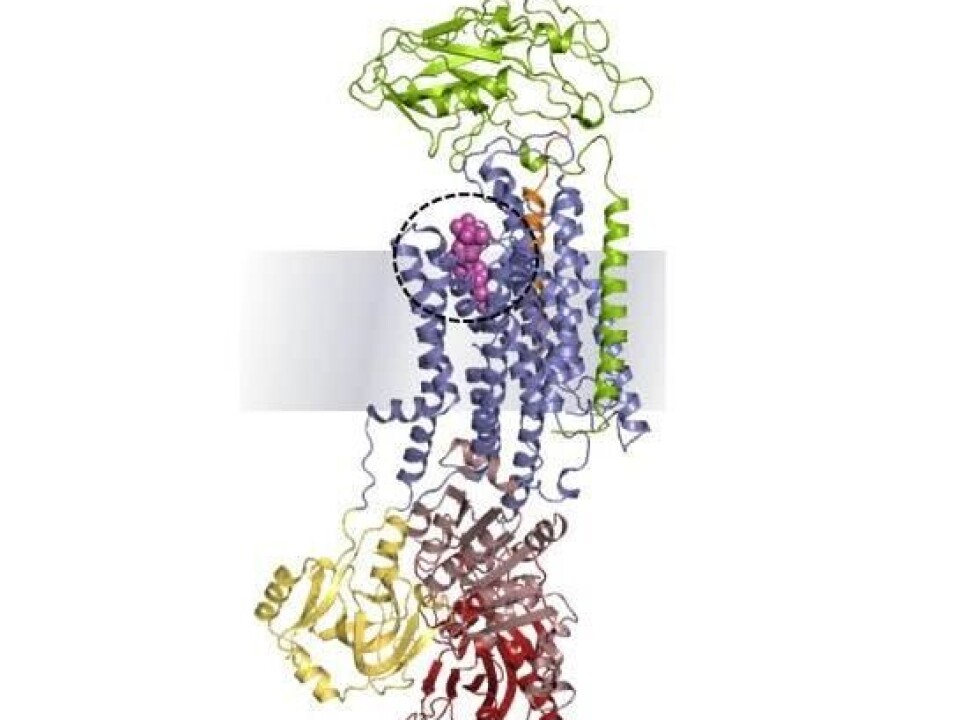
“We have, as the first scientists ever, described how the cardiotonic steroids bind to the Na+/K+-ATPase at the atomic level, so now we are armed with detailed new information, which can be used for developing new types of drugs that are specifically targeted at a number of life-threatening diseases,” says Fedosova.
From enzymes in pig’s kidneys to crystals
She and her colleagues’ description of the microscopic molecular process is published in the journal PNAS. The study is the result of years of laboratory work, which included pig’s kidneys:
”Kidney tissue contains high levels of sodium pump enzyme, so we isolated the cell membranes from pig’s kidneys. We then added a cardiotonic steroid from plants known as ouabain,” explains the researcher.
“Next, we tested a variety of additives, temperatures and concentrations to get the ouabain and the sodium pump to form a crystal. This is a complex and lengthy process.”
Crystals touring Europe
After repeated trials in the laboratory, the researchers finally managed to get the sodium pump to form crystals.
The crystals were frozen in liquid nitrogen and transported to France, Switzerland and Germany, where they have so-called synchrotrons, a type of cyclic particle accelerator which exposes the crystallised enzyme to intense X-ray radiation.
By analysing the bent X-rays from the crystal, the researchers could deduce how the atoms are placed, and after painstaking efforts, they also uncovered the three-dimensional structure of the crystallised enzyme.
“Our data shows how more than 10,000 atoms are placed in the molecules, and that enabled us to explain how the binding between the cardiotonic steroid and the sodium pump works.”
--------------------------
Read the Danish version of this article at videnskab.dk
Translated by: Dann Vinther